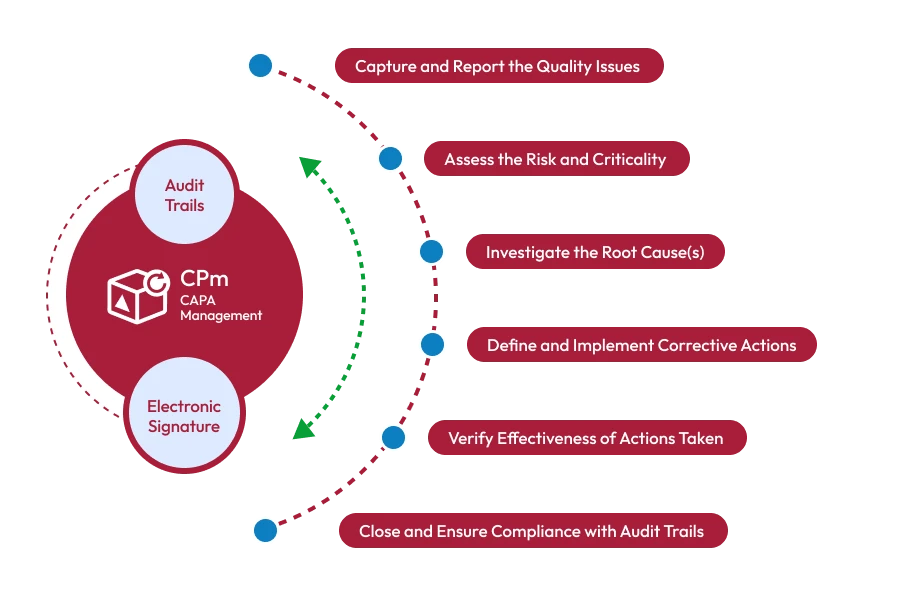
Organizations across regulated industries must constantly monitor quality processes, identify issues, and implement corrective measures to maintain product standards and regulatory compliance. Corrective and Preventive Action (CAPA) plays a crucial role in this process by ensuring that problems are addressed at their root cause and prevented from recurring.
Managing CAPA through spreadsheets or manual documentation can create inefficiencies and increase the risk of errors. Corrective and preventive action software provides a structured and automated approach to managing quality issues. Platforms such as Qualityze CAPA Software offer advanced capabilities that help organizations streamline CAPA workflows, improve compliance, and drive continuous improvement.
What Is Corrective and Preventive Action (CAPA)?
Corrective and Preventive Action (CAPA) is a systematic approach used to identify, investigate, and resolve quality issues. The process involves determining the root cause of problems and implementing corrective actions to fix existing issues while establishing preventive actions to avoid similar problems in the future.
CAPA is a core component of any quality management system and is widely used in industries such as pharmaceuticals, life sciences, manufacturing, and aerospace.
Why Organizations Need CAPA Software
As organizations grow and regulatory requirements become more complex, managing CAPA processes manually becomes difficult. CAPA software helps organizations automate quality processes and maintain complete visibility across corrective and preventive actions.
Key reasons organizations implement CAPA software include:
-
Improved tracking of quality issues
-
Faster root cause investigation
-
Automated workflows and approvals
-
Better regulatory compliance
-
Centralized documentation and reporting
By digitizing CAPA processes, organizations can reduce operational risks and ensure that quality issues are resolved effectively.
Key Features of Corrective and Preventive Action Software
Modern CAPA solutions provide powerful features that improve quality management and operational efficiency.
Automated Workflow Management
CAPA software automates the entire lifecycle of corrective and preventive actions, from issue identification to closure. Automated notifications and task assignments ensure that responsibilities are clearly defined and tracked.
Root Cause Analysis Tools
Effective CAPA management requires identifying the true cause of a problem. CAPA software provides tools such as root cause analysis, investigation tracking, and corrective action planning.
Centralized Documentation
All CAPA records, reports, and supporting documents are stored in a centralized system. This ensures easy access to information during internal reviews and regulatory audits.
Compliance and Audit Readiness
Digital CAPA systems maintain detailed audit trails and ensure that organizations comply with industry standards and regulatory requirements.
Real-Time Reporting and Analytics
Advanced dashboards provide insights into CAPA performance, helping organizations identify recurring issues and improve decision-making.
How Qualityze Enhances CAPA Management
Qualityze provides a cloud-based CAPA management solution that helps organizations manage corrective and preventive actions more efficiently.
The platform enables businesses to:
-
Automate CAPA workflows and approvals
-
Perform structured root cause analysis
-
Maintain audit-ready documentation
-
Monitor CAPA performance with real-time dashboards
-
Integrate CAPA with other quality management processes
Qualityze integrates CAPA with modules such as document control, audit management, training management, and nonconformance management, creating a unified quality management environment.
Benefits of Implementing CAPA Software
Implementing corrective and preventive action software provides several advantages for organizations.
Improved Quality Management
CAPA software ensures that quality issues are identified and addressed quickly, improving overall product and process quality.
Faster Issue Resolution
Automated workflows help teams investigate and resolve problems more efficiently.
Enhanced Regulatory Compliance
Digital documentation and audit trails make it easier to demonstrate compliance with regulatory standards.
Stronger Continuous Improvement
By analyzing CAPA data and identifying trends, organizations can implement improvements that prevent future issues.
Industries That Use CAPA Software
Corrective and preventive action software is widely used in industries where quality and compliance are critical.
These industries include:
-
Life sciences
-
Pharmaceuticals
-
Medical devices
-
Manufacturing
-
Aerospace
-
Automotive
Organizations in these sectors rely on CAPA systems to ensure regulatory compliance and maintain product quality.
Why Choose Qualityze CAPA Software?
Organizations looking for a modern CAPA solution often choose Qualityze because of its scalable cloud platform and advanced automation capabilities.
Qualityze helps businesses streamline corrective and preventive actions, improve collaboration across teams, and maintain complete visibility into quality processes. With an integrated QMS platform, organizations can ensure that quality issues are resolved quickly and effectively.
Conclusion
Corrective and preventive action software plays a critical role in modern quality management. By automating CAPA processes, organizations can improve compliance, reduce risks, and drive continuous improvement.
Solutions like Qualityze provide a comprehensive platform that helps businesses manage CAPA workflows, investigate root causes, and ensure long-term quality success.