The report "CAR T-Cell Therapy Market by Product (Abecma, Breyanzi, Carvykti, Yescarta, Tecartus, Kymriah), Target (CD19, BCMA), Indication (Multiple Myeloma, Leukemia, Lymphoma), Demographic (Adult, Pediatric), Region, Competitive Landscape - Global Forecast to 2031" The CAR T-Cell Therapy market is projected to reach USD 13.56 billion by 2031 from USD 6.78 billion in 2026, at a CAGR of 14.9% during the forecast period. The market's rapid growth is primarily driven by the increasing prevalence of cancer, rising investments in advanced immunotherapies, and continuous technological advancements in CAR T-cell treatment approaches.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=47772841
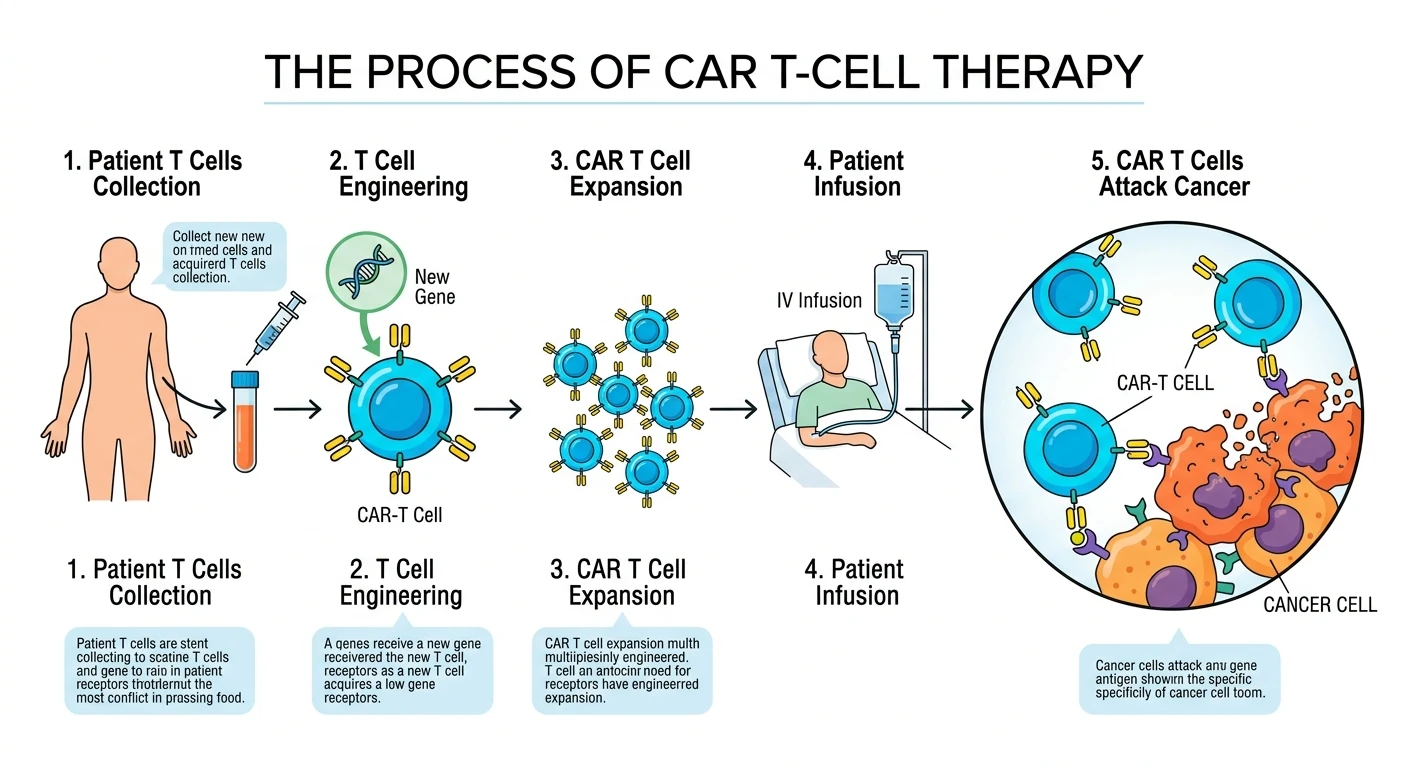
CAR T-cell therapy, a revolutionary form of personalized cancer treatment, is gaining widespread adoption due to its ability to deliver targeted and durable responses in hematologic malignancies. Growing clinical success rates, coupled with expanding regulatory approvals and ongoing research, are further accelerating market growth.
Product Segment Dominated by Carvykti
Based on product segmentation, the market includes leading therapies such as Abecma (idecabtagene vicleucel), Breyanzi (lisocabtagene maraleucel), Carvykti (ciltacabtagene autoleucel), Yescarta (axicabtagene ciloleucel), Tecartus (brexucabtagene autoleucel), and Kymriah (tisagenlecleucel). Among these, Carvykti accounted for the largest market share in 2025.
The strong adoption of Carvykti is attributed to its proven clinical efficacy in treating relapsed or refractory multiple myeloma, demonstrating deep response rates and durable remission outcomes. Furthermore, recent label expansions into earlier lines of therapy and increasing availability across certified treatment centers have significantly boosted its uptake.
B-Cell Lymphoma Leads Indication Segment
By indication, the CAR T-cell therapy market is segmented into acute lymphoblastic leukemia (ALL), multiple myeloma, B-cell lymphoma (BCL), and other indications. In 2025, B-cell lymphoma emerged as the leading segment.
This dominance is largely due to the rising incidence of B-cell lymphomas, which represent a substantial patient population eligible for CAR T-cell therapies. The expanding use of CAR T-cell treatments in this indication continues to support strong market growth.
CD19 Target Segment Holds Largest Share
Based on target type, the market is categorized into BCMA, CD19, CD19/20, CD7, and other targets. The CD19 segment accounted for the largest share in 2025.
CD19 is widely expressed on malignant B-cells, making it a highly effective and commonly targeted antigen in CAR T-cell therapies. The prevalence of CD19-positive cancers, particularly B-cell lymphomas and leukemias, has significantly contributed to the dominance of this segment.
North America Leads Global Market
Regionally, North America held the largest share of the CAR T-cell therapy market in 2025 and is expected to maintain its leadership during the forecast period.
The region's dominance can be attributed to advanced healthcare infrastructure, early adoption of innovative therapies, and strong regulatory support, including early approvals from the US Food and Drug Administration (FDA). Additionally, substantial investments in research and development and the presence of leading biopharmaceutical companies further strengthen the region's position.
Key Market Players
The CAR T-cell therapy market is highly competitive, with key players focusing on innovation, strategic collaborations, and pipeline expansion. Prominent companies operating in this space include Bristol-Myers Squibb Company (US), Gilead Sciences, Inc. (US), Novartis AG (Switzerland), Johnson & Johnson (US), and JW (Cayman) Therapeutics Co. Ltd (China). These companies are actively advancing next-generation CAR T-cell therapies to enhance efficacy, safety, and accessibility.